 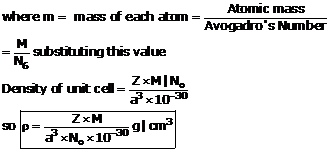
Typically, the fuel has an enrichment of ω 235 = 4% of isotope 235U.Ĭalculate the atomic number density of 235U (N235U), when: Most of PWRs use uranium fuel, which is in the form of uranium dioxide (UO 2). 
The atomic number density of isotope i is: The enrichment may be specified in terms of weight percent or weight fraction, ω i, of isotope i: In nuclear engineering, we are working with the term fuel enrichment. N C = 2.75×10 22 atoms of 12C/cm 3 Example – Fuel enrichment N C = 1 x 2.75×10 22 atoms of carbon/cm 3 
Boron carbide has a density of 2.52 g/cm 3.ĭetermine the atomic number densities of individual constituents. Natural boron consists primarily of two stable isotopes, 11B (80.1%) and 10B(19.9%). Atomic Number Density of Mixtures and Compoundsįor a chemical compound (mixture) Z or for an isotopic mixture, which is composed of elements or isotopes X and Y, the number (atom) density of the compound is calculated from:Įxample – Atomic number densities of boron carbideĪ control rod usually contains solid boron carbide with natural boron. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
